Time:2020-06-16
A recent study published in American Journal of Psychiatry reported a novel cross-species translational approach for diagnostic classification in human mental disorders. This work was performed by researchers in Dr. WANG Zheng’s Lab at the Institute of Neuroscience, Center for Excellence in Brain Science and Intelligence Technology, in close collaboration with Dr. HE Ran’s group at the Institute of Automation of the Chinese Academy of Sciences.
ASD represents a heterogeneous group of neurodevelopmental disorders and often occurs with co-morbidities such as obsessive-compulsive disorder (OCD) and attention deficit–hyperactivity disorder (ADHD). The substantial phenotypic heterogeneity and high frequency of co-morbidity present a formidable challenge for accurate diagnosis. The success of new gene-editing technologies has stirred a growing interest in the use of nonhuman primates to investigate the biomechanisms of brain diseases. For instance, MECP2 gain- and loss-of-function in genetically-engineered monkeys have demonstrated typical phenotypes in autism such as less active social contact, increased stereotypical behaviors and elevated anxiety. Animal models with a clear genetic basis provide valuable opportunities for probing the gene-circuit-behavior mechanisms, and promoting development and preclinical testing of new therapeutics.
Considering the evolutionarily conserved features of the brain connectome across primate species, researchers proposed that a subset of brain regions that particularly relevant to the core neuropathology of autism are potentially useful for cross-species mapping in the present framework (Figure A). Thus, a machine learning algorithm was deployed to identify a set of 9 core regions based on functional connectome data in the monkey cohort (Figure B). After one-to-one mapping of these core regions to the human brain, functional connections between them were extracted and tested in four independent human cohorts: Autism Brain Imaging Data Exchange (ABIDE)-I (N = 1112), ABIDE-II (N = 1114), ADHD (N = 776) and OCD (N = 186). The sparse logistic regression classifiers were trained for discriminative classification of subjects in each individual cohort. After cross-validation, the monkey-derived classifier achieved an accuracy of 82.14% in the ABIDE-I cohort, and an accuracy of 75.17% in the ABIDE-II cohort, both of which significantly outperformed the human-derived classifiers (Figure C). Moreover, the same set of core regions was useful for improved classification in the OCD cohort (78.36%), but not in the ADHD cohort. Further analysis of these superior monkey-derived classifiers revealed that distinct connections from the ventral lateral prefrontal cortex predicts differential dimensional symptom severity of ASD and OCD, suggesting dual neuropathology-dependent mechanistic roles in these two diseases (Figure D). In short, specific brain regions or circuits derived from genetically-edited animal models may serve as a basis to define a biologically-defined circuit endophenotype cut across current diagnostic categories.
This work entitled “Diagnostic Classification for Human Autism and Obsessive-Compulsive Disorder based on Machine Learning from a Primate Genetic Model” was published in American Journal of Psychiatry on June 17, 2020. This work was completed by ZHAN Yafeng and WEI Jianze under the supervision of Dr. WANG Zheng, and Dr. HE Ran with the help of Dr. Trevor Robbins at Cambridge University, and Dr. XU Xiu at Children’s hospital of Fudan University. This work was supported by Chinese Academy of Science, National Natural Science Foundation, Shanghai City, Guangdong Province, and the Wellcome Trust.
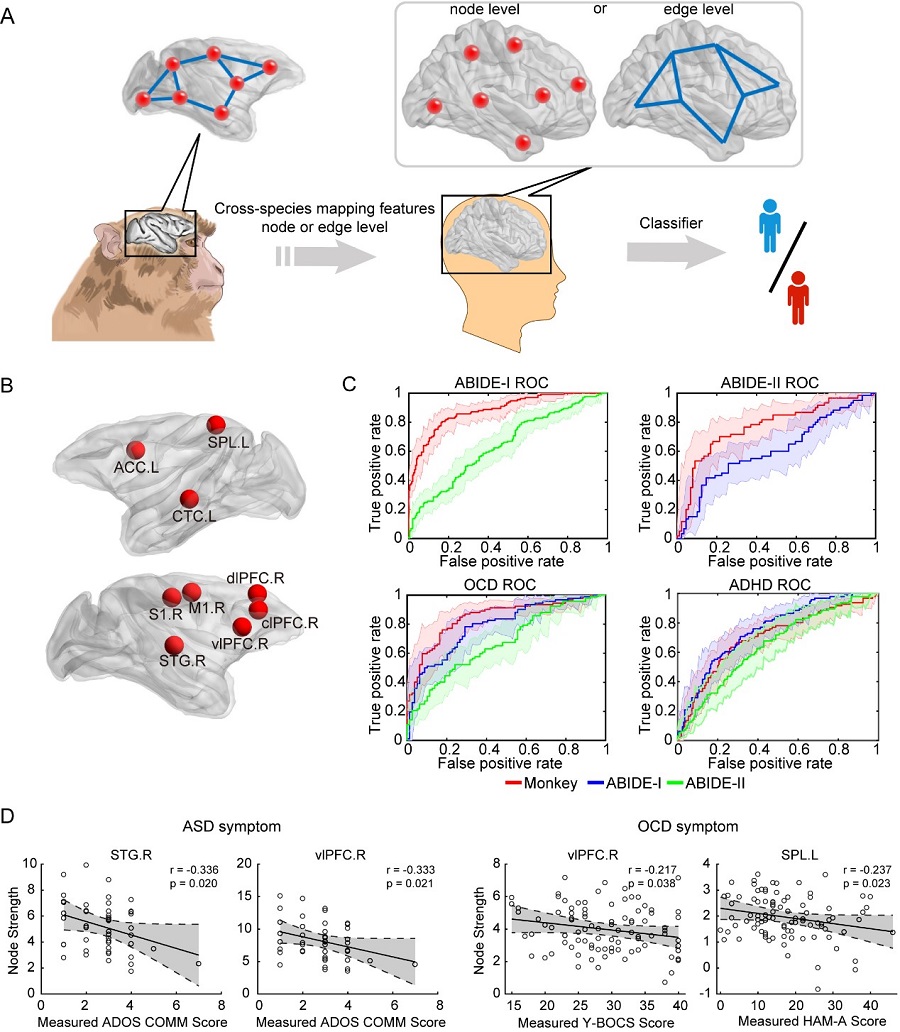
Figure legend: (A) Illustration of a cross-species circuit mapping strategy, which is formulated on the basis of the intuition that characteristics of the brain circuitry between nonhuman and human primates are evolutionarily conserved at the node or edge level. (B) Spatial illustration of 9 core regions identified from the monkey cohort. (C) Receiver operating characteristic (ROC) curves of the monkey-derived classifier are plotted against those of the human-derived classifiers. (D) Node strength of specific regions or neural circuits that are associated with differential behavioral domains in ASD and OCD. (Image by CEBSIT)
 附件下载:
附件下载: