Time:2020-05-18
A recent study published in Cell Reports demonstrates that retinoid X receptor α (Rxra) regulates docosahexaenoic acid (DHA)-dependent spinogenesis and functional synapse formation in vivo. This work was performed by researchers in Dr. YU Xiang’s Laboratory at the Institute of Neuroscience, Center for Excellence in Brain Science and Intelligence Technology of Chinese Academy of Sciences, State Key Laboratory of Neuroscience. Using a combination of gene expression manipulations (transgenic mice, viral injections and in utero electroporation), pharmacology, immunohistochemistry, RNA-Seq, fluorescence dye microinjection, neuronal morphology analyses, slice electrophysiology and behavioral tests, they identified an Rxra-dependent unesterified DHA signaling pathway required for functional synapse formation, providing insight into the mechanism through which DHA promotes brain development and cognitive function.
The formation of functional synaptic connections is critical to neural circuit wiring. During early development, synapse formation predominates, leading to a rapid increase in total synapse number. Synaptogenesis regulated by coordinated extracellular cues and intracellular signaling. Nuclear receptors are ligand-activated transcription factors that regulate development, homeostasis, and metabolism of the organism. However, relatively little is known about how nuclear receptors regulate synapse formation. Rxra is a nuclear receptor that can function as both homo- and heterodimers. DHA, a brain-enriched polyunsaturated fatty acid and important nutritional supplement in baby formula, has been proposed to be the endogenous ligand of RXRA, although direct in vivo evidence is lacking. DHA level in the brain raises rapidly during early development, plateauing in adulthood, following a time course similar to that of synaptogenesis.
Evidence from both human and animal studies suggest that DHA is critical to brain development and function. Long-term DHA deprivation (weeks or months) in vivo has been shown to impair synapse function and cognition-related performance, while long-term DHA supplementation improved both parameters. However, the mechanism by which DHA regulates synapse function and/or cognition-related performance remains largely unknown. In contrast to the long period needed to induce in vivo changes, in vitro administration of unesterified DHA promoted synapse development on the time scale of hours to days. Based on existing knowledge, the authors hypothesized that unesterified DHA may regulate synapse development through activation of its receptor RXRA.
The authors first examine the contributions of RXRA and DHA as a potential ligand-receptor pair, to the processes of spinogenesis and functional synapse formation in the cerebral cortex. They found significantly reduced dendritic spine density of pyramidal neurons in both developing and adult sensory cortices, following conditional knockout (cKO) of Rxra from these neurons. They further showed that the effects of RXRA are mediated through its DNA-binding domain, in a cell-autonomous and reversible manner. The morphological changes were accompanied by reduced excitatory synaptic transmission.
Importantly, intracerebroventricular (i.c.v.) injection of unesterified DHA into the brain significantly upregulated spine density and excitatory synaptic transmission in wildtype mice, an effect blocked in Rxra cKO mice. Blocking DHA release from brain phospholipids led to significant reduction in spine density, an effect rescued by supply of unesterified DHA. DHA and RXRA regulated the expression of immediate early genes (IEGs), important regulators of synapse formation. Finally, they found that Rxra cKO mice exhibited impairments in behaviors associated with social memory.
In summary, Dr. Yu’s lab demonstrated that unesterified DHA signals through RXRA to regulate spinogenesis and functional synapse formation, providing insight into the mechanism through which DHA promotes brain development and cognitive function.
This work entitled “Retinoid X Receptor α Regulates DHA-dependent Spinogenesis and Functional Synapse Formation in vivo” was published online in Cell Reports on May 19, 2020. This work was completed by CAO Huateng and LI Minyin, under the supervision of Dr. YU Xiang, with help from LI Guangying, LI Shujing, HUANG Shajin, LU Yuan and WEN Bincheng. This work was supported by grants from the Ministry of Science and Technology of China, National Natural Science Foundation of China, Chinese Academy of Sciences, and Shanghai Municipal Government.
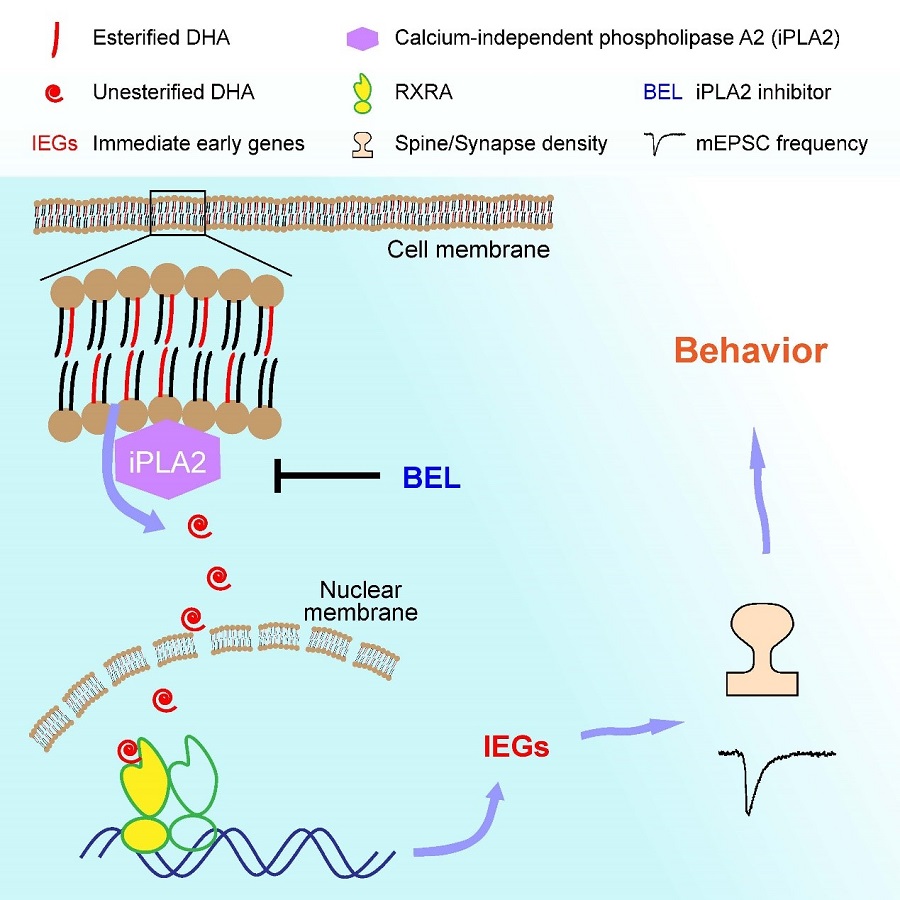
Figure legend. A model illustrating how unesterified DHA and RXRA regulate spinogenesis and synaptic transmission. Upon activation of phospholipase (iPLA2), unesterified DHA is released from the membrane, enters into the nucleus and binds to the nuclear receptor RXRA, resulting in enhanced IEG expression, and in turn promoting spinogenesis and synaptic transmission, eventually resulting in behavioral changes.(Image by CEBSIT)
AUTHOR CONTACT: YU Xiang
Senior Investigator, Institute of Neuroscience, Center for Excellence in Brain Science and Intelligence Technology of Chinese Academy of Sciences
320 Yue-Yang Road, Shanghai 200031, China
Email: yuxiang@ion.ac.cn 附件下载:
附件下载: