Time:2020-04-16
A recent study published in Science Advances reports a highly sensitive and selective potassium ion (K+) nanosensor that can be excited by near-infrared (NIR) light. This work was performed by researchers in Dr. DU Jiulin’s and Dr. XIONG Zhiqi’s Labs at the Center for Excellence in Brain Science and Intelligence Technology, Institute of Neuroscience and State Key Laboratory of Neuroscience, and in Dr. SHI Jianlin’s and Dr. BU Wenbo’s Labs at State Key Laboratory of High Performance Ceramics and Superfine Microstructure, Shanghai Institute of Ceramics, Chinese Academy of Sciences.
K+ is an essential component of neural activity. The difference between its intracellular and extracellular concentration affects neuronal activation and synaptic transmission, and changes itself upon spiking activities. Despite the demand for a selective K+ sensor, the previous efforts in developing such sensors resulted in limited satisfaction in differentiating K+ from sodium ion (Na+). For reporting K+ concentration, non-invasive fluorescence detection of K+ holds great potential to convey the spatial and temporal information about [K+]. Thus a fluorescent K+-selective sensor is required to unravel the role of K+ in complex neural interactions across various scales in the brain.
The new nanosensor was created by encapsulating upconversion nanoparticles (UCNPs) and a commercial K+ indicator in the hollow cavity of mesoporous silica nanoparticles, followed by coating it with a layer of K+-selective filter membrane. K+-permeable membrane filter on the nanosensor is effective at filtering out other cations and capturing K+ ions exclusively. The UCNPs convert NIR to ultraviolet light, which excites the K+ indicator, thus it allowed the detection of the fluctuations of [K+] in cultured cells and intact brain of mouse and zebrafish under infrared illumination.
To demonstrate the wide application of this new nanosensors, the researchers used it in a migraine mouse model and epilepsy zebrafish model. Cortical spreading depression (CSD) has been hypothesized to be the underlying mechanism of the migraine aura. The CSD, a slowly propagating wave of depolarization followed by brain activity suppression, is remarkably complex and involves dramatic changes in both membrane potential and [K+]. Although accurate measurement of [K+] is possible using microelectrodes, their fabrication is technically challenging and their deployment involves brain invasion. Furthermore, from one or two single electrodes, it’s difficult to obtain the spatial and temporal aspects of [K+]. By using the newly developed nanosensor in the mouse brain, the authors observed that a wave of increasing [K+] propagated gradually across the cortex, and provided a new window to explore the mechanism underlying the CSD.
While the initiation of epilepsy is still in debate, it was suggested that defects in extracellular potassium buffering and the thus accumulated potassium may facilitate epilepsy initiation and propagation by elevating neuronal activation. To test this possibility, we simultaneously monitored the neuronal activity and the extracellular potassium concentration by deploying the newly developed nanosensor in the brain of zebrafish larvae panneuronally expressing red calcium sensor. Upon epilepsy induction, both neuronal calcium activity and extracellular potassium concentration increased widely. However, potassium accumulated beyond regions with elevated neuronal activity, suggesting that potassium increase did propagate away from the releasing loci, consistent with its possible contribution in epilepsy initiation and propagation.
This study successfully demonstrated that the developed nanosensor construction strategy would contribute not only to scientific discoveries and breakthroughs in neuroscience research, but also to the development of other selective ion sensors.
This work entitled “A highly sensitive and selective nanosensor for near-infrared potassium imaging " was published online in Science Advances on April 18th. LIU Jianan, PAN Limin, and SHANG Chunfeng contributed equally as the first authors. Dr. DU Jiulin, Dr. XIONG Zhiqi, Dr. SHI Jianlin, and Dr. BU Wenbo are corresponding authors. It was financially supported by China Association for Science and Technology, Chinese Academy of Sciences, Science and Technology Commission of Shanghai Municipality, Guangdong Innovation Platform of Translational Research for Cerebrovascular Diseases.
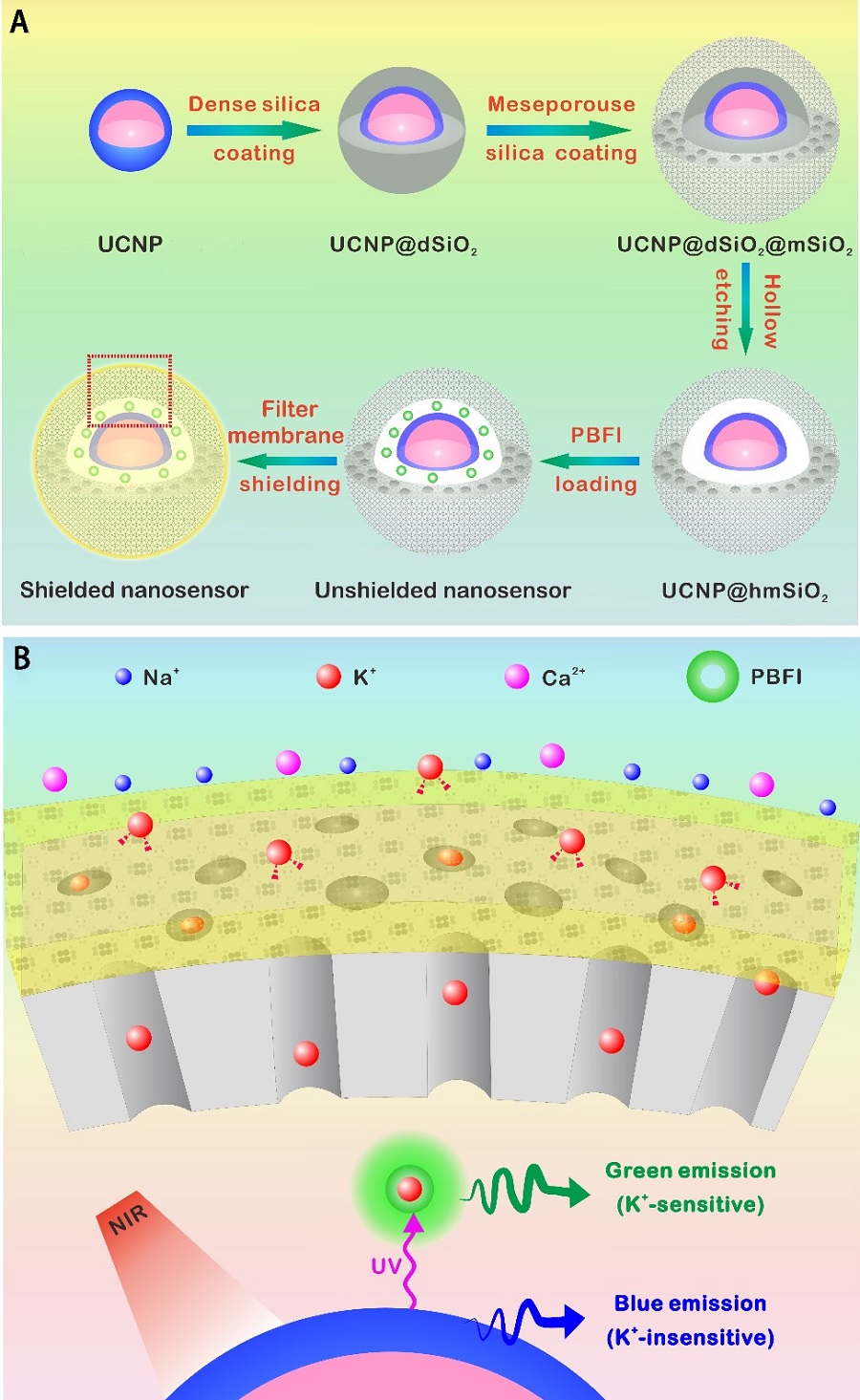
Figure legend. Design and sensing mechanism of the K+ nanosensor. (A) Schematic illustration for the synthesis of the nanosensor. The NaYF4:Yb/Tm@NaYF4:Yb/Nd (UCNP) core was synthesized and coated with a dense silica layer and a successive mesoporous silica shell. Etching away of the dense silica layer forms a hollow cavity that allows the loading of PBFI. The nanosensor was lastly coated with the K+-selective filter membrane. (B) Schematics showing a magnified view of the nanosensor [from the red dotted box in (A)] and its K+ sensing mechanism. The filter membrane layer allows only K+ to diffuse in and out of the nanosensor, thus excluding the interference from other cations. Once diffused into the nanosensor, K+ will bind to PBFI immediately. Upon NIR irradiation, the upconverted UV light from the UCNPs excites PBFI, leading to the emission of K+-bonded PBFI. (Image by CEBSIT)
 附件下载:
附件下载: