Time:2023-11-22
A recent study published in Immunity demonstrated that intestinal epithelial DRD2 specifically promotes Experimental autoimmune encephalomyelitis (EAE) in female mice by altering the composition of the intestinal flora and its metabolites. This work was performed by researchers in Dr. Zhou Jiawei’s Lab at the Institute of Neuroscience, Center for Excellence in Brain Science and Intelligence Technology of the Chinese Academy of Sciences (CAS), State Key Laboratory of Neuroscience, Dr. Xinyang Song’s Lab at the Center for Excellence in Molecular Cell Science, CAS; Dr. Zhu Zhengjiang’s Lab at the Interdisciplinary Research Center on Biology and Chemistry, Shanghai Institute of Organic Chemistry, CAS and Dr. Chen Sheng’s team at Ruijin Hospital, affiliated to the Shanghai Jiaotong University School of Medicine. This work has indicated that activation of the intestinal epithelial dopamine signaling pathway increases the susceptibility of females to central nervous system (CNS) autoimmune diseases, laying a foundation for sex-specific interventions for these diseases.
CNS autoimmune diseases including multiple sclerosis (MS), neuromyelitis optica and neuromyelitis optica spectrum disorders, are characterized by chronic and progressive neuroinflammation, demyelination and neurodegeneration. These diseases show a strong female predisposition in terms of incidence and clinical features. Most MS patients are young and middle-aged and gradually lose their voluntary mobility as the disease progresses, and the available therapeutic agents are mostly symptomatic, with limited and expensive options, causing a huge financial and caregiving burden on families and society. Therefore, there is an urgent need in this field to develop drugs that can safely and effectively treat MS.
Alterations of the gut microbiota play a key role in autoimmune diseases. In MS patients, dysregulation of the gut microbiota affects the progression of multiple sclerosis. In animal models of multiple sclerosis, the gut microbiota plays a key role in the initiation, effector, and modulatory phases of the disease process. However, a consistent microbiota phenotype has not been established due to the wide inter-individual variation in the composition of the gut microbiota. The intestinal epithelial cells constitute the monolayer of the gastrointestinal tract, which not only insulates the intestinal lumen and its contents, but also integrates the signals of multiple microbiota within the intestinal lumen to maintain normal physiological function of the gastrointestinal tract. Whereas a variety of MS-associated intestinal bacteria have been reported to produce dopamine receptor agonists, there is an urgent need to investigate whether intestinal epithelial dopamine receptors mediate microbiota and host communication with each other as well as their roles in MS.
In order to study the effect of intestinal epithelial dopamine receptors on multiple sclerosis, Zhou’s Lab constructed mouse lines lacking dopamine D2, D3, and D4 receptor in intestinal epithelial cells , respectively. Phenylethylamine, which is produced in large quantities by intestinal bacteria, was selected as an agonist of dopamine receptors according to a previous study. EAE was chosen as an animal model for MS. Dopamine receptor knockout mice were examined for systemic changes in EAE, such as EAE clinical score, pathologic analysis, and multi-omics analysis. The identified differential metabolites were screened for their activities for suppression of inflammatory response in microglial cell lines and capabilities to alleviate EAE in vivo . Feces from MS patients were also collected for targeted metabolite assays to verify the correlation between phenylethylamine levels and gender differences in MS pathogenesis.
First, by mass spectrometry analysis, they found higher levels of phenylethylamine in the feces of MS patients than in healthy controls, with gender differences. Then, by conditional knockout and pharmacological stimulation, they found that the intestinal epithelial dopamine receptor D2, but not D3 and D4, played a key role in the progression of autoimmune encephalomyelitis. By comparison with the transcriptome of wild-type controls, they found that DRD2 knockout mice displayed significantly reduced expression of antimicrobial peptides, such as lysozyme.A significant difference in the composition of the gut microbiota between genotypes of female mice was also detected by sequencing of the 16s bacterial flora. By co-housing and antibiotic treatment, they found that the role of DRD2 in EAE was microbiota-dependent. Up to 47 metabolites that differed only in the spinal cord of female mice were identified by an untargeted metabolomic assay. Subsequently, N-acetyl-lysine was found to significantly inhibit the inflammatory response in vivo and in vitro thereby mitigating the onset and progression of autoimmune encephalomyelitis.
To further investigate the molecular mechanism of inflammation inhibition by N-acetyl-lysine, microglia in the spinal cord were isolated and subjected to transcriptome sequencing and single-cell sequencing. Zhou’s Lab found that N-acetyl-lysine treatment decreased the proportion of MS-associated microglia and increased the proportion of proliferative and homeostatic microglia. This suggests that N-acetyl-lysine facilitates the restoration of CNS immune homeostasis that is imbalanced during EAE.
In this project, Zhou’s Lab together with Song’s Lab, Zhu’s Lab and Chen’s team identified the phenylethylamine-IEC DRD2-lysozyme axis as a critical factors that influences sex bias in EAE/MS, thus providing a molecular and cellular basis for understanding how microbiota-gut-brain interactions regulate CNS autoimmune diseases. Further characterization of the role of N-acetyl0lysine in limiting microglia activation in a mouse model of MS would provide potential therapeutic opportunities to combat microglia-mediated neuroinflammation.
This work entitled “Intestinal epithelial dopamine receptor signaling drives sex-specific disease exacerbation in a mouse model of multiple sclerosis” was published online in Immunity on November 22, 2023. Hai-rong Peng, Jia-qian Qiu, Qin-ming Zhou and Yu-kai Zhang are the first authors with equal contribution. This work was supported by CAS, MOST, Shanghai municipal government, NSFC and Science and Technology Department of Guangdong Province .
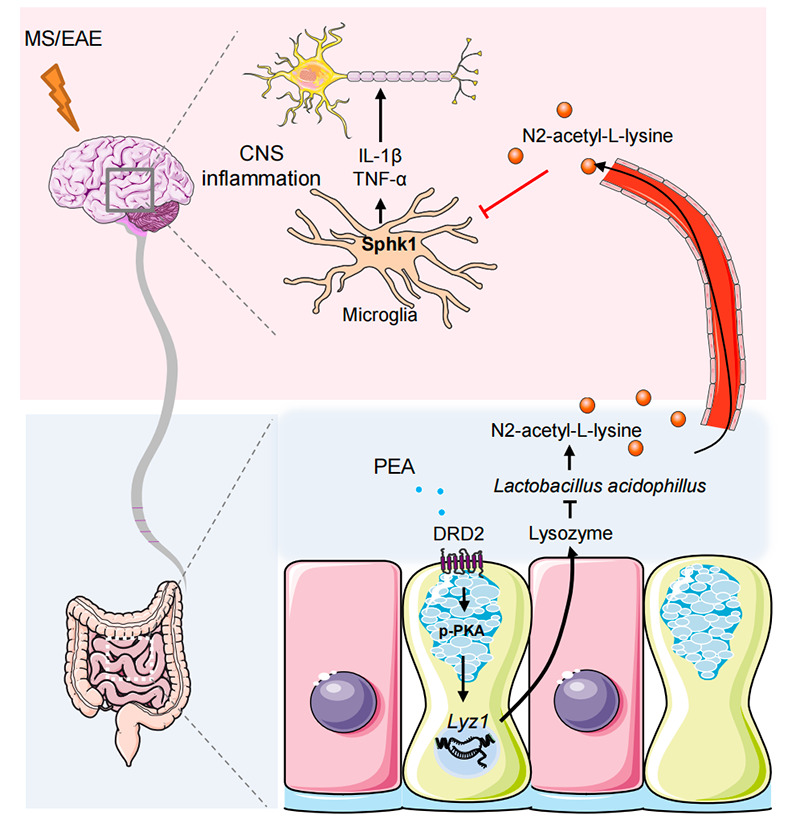
Figure 1.In female mice, higher concentrations of phenylethylamine in the intestine lead to hyperactivation of DRD2 in the intestinal epithelium, which results in increased expression of lysozyme and defensins. As a result, lysozyme-sensitive strains such as Lactobacillus are reduced in female mice. N-acetyllysine produced by Lactobacillus has a strong inhibitory effect on microglia-mediated inflammation, thereby alleviating CNS autoimmune diseases such as multiple sclerosis.
Keywords :CNS autoimmune diseases, Multiple sclerosis, Phenylethylamine, Intestinal epithelium, Dopamine D2 receptor, Lysozyme, N2-acetyl-L-lysine, Sex difference.
AUTHOR CONTACT:
Jiawei Zhou
Institute of Neuroscience, Chinese Academy of Sciences, Shanghai, China.
Phone: 13918299495; E-mail: jwzhou@ion.ac.cn
 附件下载:
附件下载: