Time:2022-06-08
A recent study published in Cell Metabolism reported a specific cluster of neurons in ventromedial hypothalamus (VMH) expressing preprodynorphin(Pdyn) could control homeostatic thermogenesis and social interaction- associated hyperthermia. This work was accomplished by Dr. ZHANG Zhe’s lab, Dr. LIANG Zhifeng’s lab and Dr. XU Xiaohong’s lab at the Institute of Neuroscience, Center for Excellence in Brain Science and Intelligence Technology of the Chinese Academy of Sciences, State Key Laboratory of Neuroscience. This research identified Pdyn-expressing neurons in ventromedial hypothalamus as a novel nucleus that not only responds to cold temperature in the environment and promotes homeostatic thermogenesis, but also modulates social interaction and associated thermogenesis.
Birds and mammals are endotherms, which could maintain the body temperature within a relative stable range despite the change of environmental temperature. A proper and stable body temperature ensures efficient metabolism, which is important for survival, as a few degrees’ change of body temperature could produce severe debilitating effects. While previous research has identified some neural pathways critical for body temperature homeostasis, the exact cell types that control thermoregulation remain elusive.
Early studies of the temperature regulation neural circuit showed that the environment temperature was sensed by the thermo-receptors that separated on the skin and this information was transmitted to the hypothalamus through spinal cord and midbrain. Pacap (Pituitary Adenylate Cyclase Activating Polypeptide) / BDNF (brain-derived neurotrophic factor) – expressing neurons in POA respond to warm environment temperature and mediate the decrease of body temperature (Tan et al., 2016). And activation of Adcyap1 (Adenylate Cyclase Activating Polypeptide 1) – expressing or Qrfp (pyroglutamylated RFamide peptide) – expressing hypothalamic neurons is capable of inducing extreme hypothermia, leading to a state of hibernation (Takahashi et al., 2020) or torpor (Hrvatin et al., 2020). While recent studies identified the BRS3 (Bombesin-like receptor 3) – expressing neurons in POA are capable of inducing thermogenesis (Pi?ol et al., 2021), and the hypothalamus-projecting glutamatergic prefrontal neurons mediate stress-induced hyperthermia (Kataoka et al., 2020), the cell types involved in cold-sensing function outside of POA remain largely unknown.
In order to systematically screen for brain regions sensitive for environment temperature, the researchers applied an alternating hot or cold stimuli to head-restrained mice and monitored brain activities using a fMRI scanner, and founded that VMH exhibited the most robust cold-responding BOLD signals (Figure A). After the analysis of single cell transcriptome data and fluorescence in situ hybridization, the researchers further identified Pdyn-expressing neurons in VMH could be the candidate population for cold sensitive neurons. The team members then used fiberphotometry to record the calcium activity of VMHPdyn neurons and verified these neurons are sensitive to the cold stimulus (Figure B). Subsequent optogenetic manipulation showed that the activation of these neurons increases body temperature, while inhibition leads to body temperature reduction (Figure C).
Using rabies virus-mediated retrograde mono-transsynaptic tracing, investigators found that VMHPdyn neurons receive a wide range of inputs from the whole brain, including Dorsal-medial Hypothalamus (DMH), Preoptic Area (POA), parabrachial nucleus (PBN) and Periaqueductal Gray (PAG). These projections indicate VMHPdyn neurons may be involved in other sensory and cognitive information together with information for regulating body temperature. Previous work reported an increase of body temperature in mice during social behavior, investigators thus recorded the calcium activity of VMHPdyn neurons and the change of body temperature simultaneously and observed an concurrent increase of both VMHPdyn neurons activity and body temperature during the social interaction. In addition, optogenetic inhibition of VMHPdyn neurons suppressed the elevation of body temperature and the social interaction (Figure D), indicates that the activation of VMHPdyn neurons could serve dual functions of modulating both social interactions and thermogenesis.
In this project, ZHANG’s lab, LIANG’s Lab and XU’s Lab identified VMHPdyn neurons are critical to maintain the body temperature in the cold environment which add an important piece to the neural circuit for thermoregulation. What is more, this work discovered a dual function of VMHPdyn neurons that coordinate energy metabolism and social behaviors.
This research entitled “Cold-sensitive ventromedial hypothalamic neurons control homeostatic thermogenesis and social interaction associated hyperthermia” was published online in Cell Metabolism on June 7, 2022. FENG Chenzhang, WANG Ying and ZHA Xi are the first authors. This work was supported by CAS and NSFC.
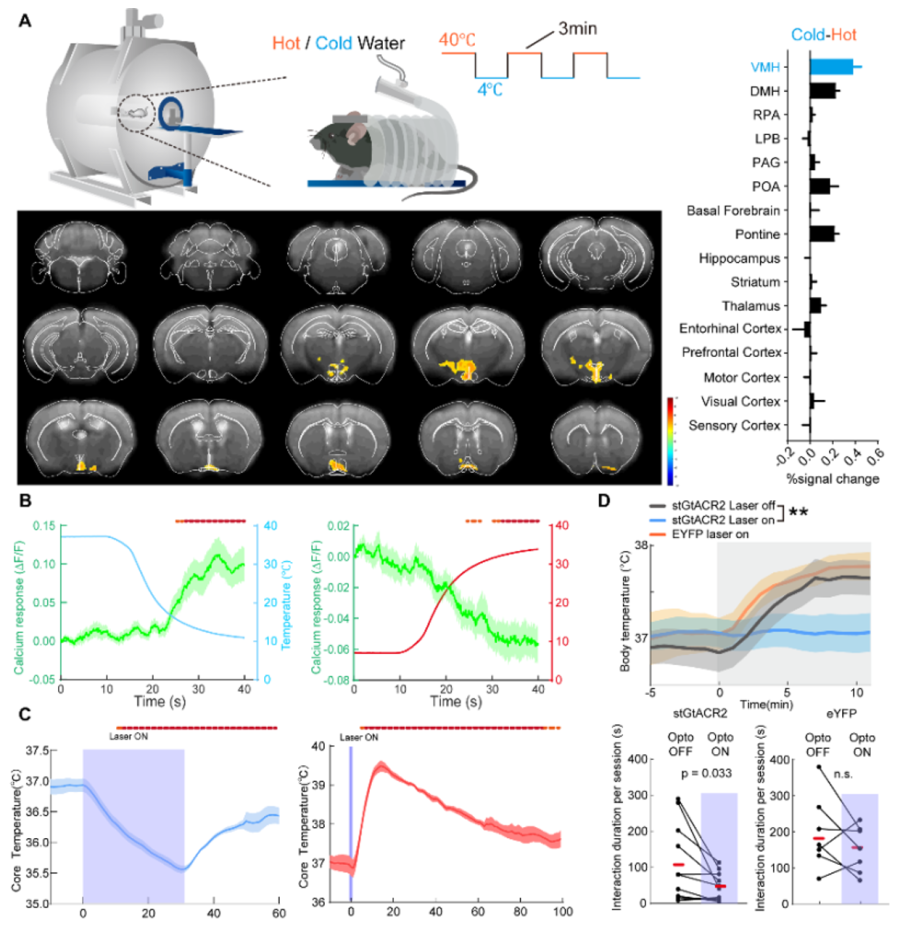
(A) Figure legend: (A) left top:Schematics of the fMRI-scanning setup for thermal challenge tests. left bottom: Representative cold-warm activation map generated through alternating thermal conditions. right: Quantification of average BOLD responses differences between cold and warm stimulation in a list of brain regions. (B) Mean calcium trace (green) during the first 30 seconds of cold or hot stimulation. Temperature stimulation was represented as blue curve (for cold) or red curve (for hot).. (C) Average traces for physiological changes with optogenetic manipulations of dmVMHPdyn neurons (D) Top: Quantification of body temperature during social interaction with optogenetic inhibition of dmVMHPdyn neurons. Bottom: Quantification of social investigation duration per session with (purple shade) or without optogenetic inhibition of dmVMHPdyn neurons.
Keywords: Ventromedial hypothalamus; Cold-sensitive; Homeostatic thermogenesis; social interaction associated hyperthermia; Preprodynorphin
AUTHOR CONTACT:
ZHANG Zhe
Center for Excellence in Brain Science and Intelligence Technology, Chinese Academy of Sciences, Shanghai, China.
E-mail: zhezhang@ion.ac.cn
 附件下载:
附件下载: